salesforce for roche
Implementing product management for Roche with Salesforce Sales Cloud

Duration of project: January 2022 - Jun 2022
Executive summary
Roche is leading pharmaceutical company, operating complex product distribution across European markets. When I joined the project, their inventory was fragmented across independent systems. The result was a manual, operational model that created significant amount of extra work for specialist and compliance.
The objective was to consolidate Roche’s product lifecycle into a single Salesforce ecosystem. From the point of sale through maintenance, replacement, and fault resolution, without manual handoffs between departments.
I led the project as the UX and product design principal, operating within an Agile framework across a cross-functional team that included Roche’s legal and compliance department, pharmaceutical specialists, Salesforce engineers, and project managers. I had design ownership, from discovery to delivery, within a six-month timeline.
Discovery & strategic insights
With no existing architecture, I orchestrated a discovery programme with multiple stakeholder groups including pharmaceutical staff, sales specialists, and distributors. The goal was to map the complete product lifecycle and locate every point where manual intervention was required.
The legacy environment forced staff into multi-system workflows for tasks that can be automated. A representative selling an asthma pump required a specialist to register the sale in one system, then manually log the customer in a separate system to trigger the three-month replacement cycle.
Working directly with Roche’s legal department, I mapped the full regulatory requirement against the proposed Salesforce architecture. The design challenge was to make compliance data available without making it intrusive, present for auditors when needed, invisible to medical staff during daily workflows.
Reducing risk through discovery sprints
Because we were building without an existing reference, validation was critical. I led Lean discovery sprints and usability testing with pharmaceutical staff and Roche distributors, stress-testing the proposed logic and inventory flows against real-world scenarios before committing. This validation surfaced edge cases in the logic early, when the cost of correction was low.
The solution
Unified product model
The foundation was a single architecture that eliminated manual handoffs entirely. Rather than managing sales and operations in separate systems, I designed the Salesforce platform so that each transition was triggered by system logic.
Three stages were fully automated:
- Point of Sale: Registration of a sale automatically triggers the support and replacement lifecycle for that specific customer and product unit
- Maintenance Triggers: Routine replacement schedules and service intervals are initiated by system logic on a defined cadence, no manual scheduling required
- Fault Management: Exchange and replacement workflows for faulty units route through automated logic.
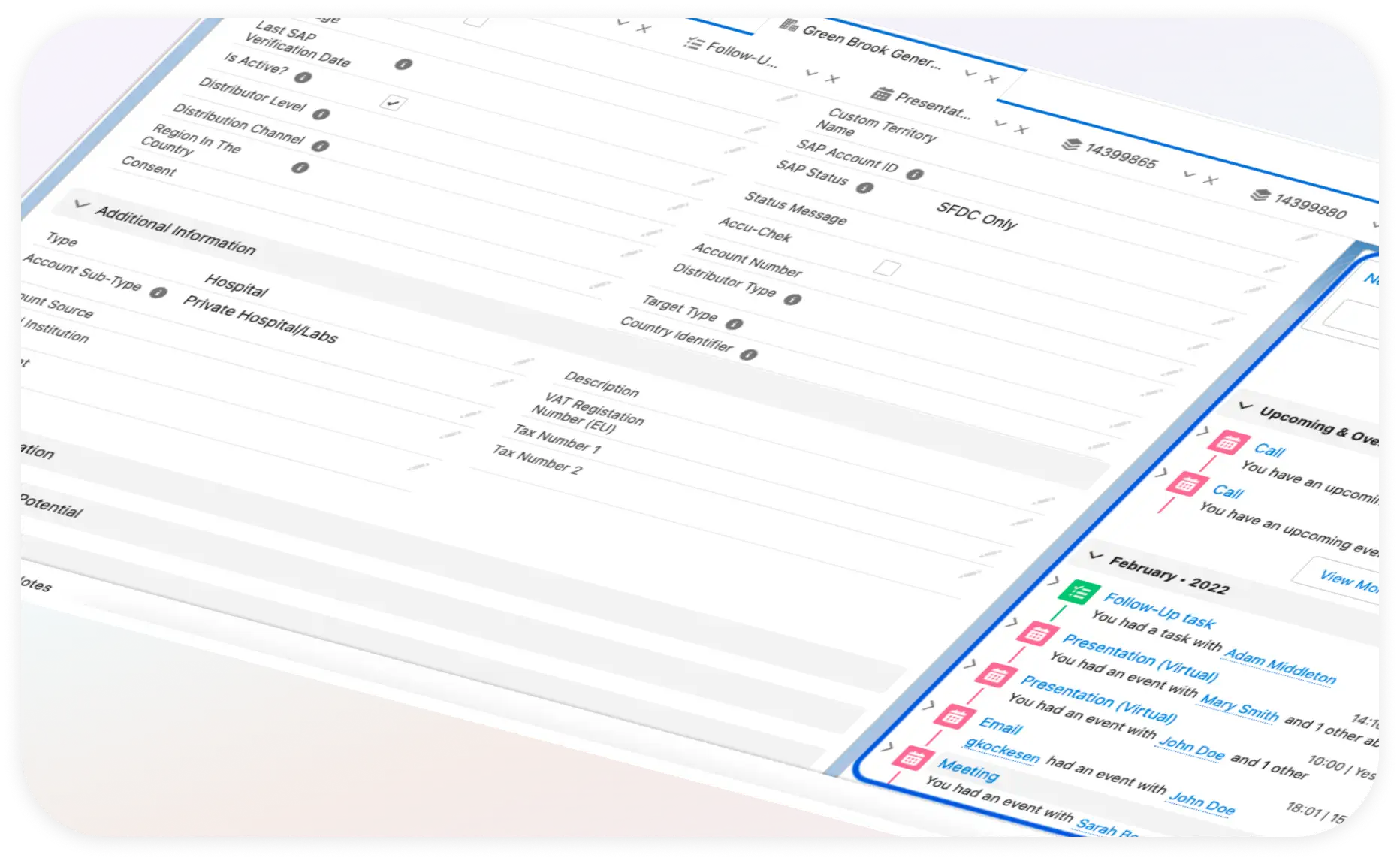
This solution removed the multi-system dependency, compliance risk, and operational error across.
This decision was validated with medical staff and Roche’s legal team before implementation, ensuring the design served both audiences.
Impact & business value
The six-month engagement delivered full-scope migration without data loss or disruption.
Quantifiable outcomes
- 100% of Roche’s product migrated into the new Salesforce Sales Cloud architecture, with no data loss across the transition
- 100% completion rate for all customer requests and technical requirements defined in the project scope
- Replacement cycle removed hours of weekly manual scheduling
Qualitative outcomes
- Staff reported high satisfaction with the automated workflow, specifically citing the elimination of manual data re-entry that had previously consumed significant time across the operation